Over my 15 years on the production floor, I’ve watched the global nutraceutical contract manufacturing industry transition into a $182.47 billion economic powerhouse, projected to reach $386.6 billion by 2033 [1]. My team and I see the drivers of this growth daily: a structural shift in consumer behavior toward preventive healthcare, personalized nutrition, and clean-label transparency, which has created a volatile operational environment for brand owners.
The days of cheaply outsourcing white-label products overseas are over. Today, brands must navigate complex regulatory frameworks (like DSHEA and 21 CFR Part 111), survive stringent new third-party testing mandates from major e-commerce platforms, and adapt to shifting consumer demands for high-absorption delivery formats. Failing to adapt to these shifts, such as getting caught in Amazon’s strict TIC verification portals, can bankrupt an unprepared brand overnight.
This definitive guide deconstructs the operational, financial, and scientific mechanics of contract manufacturing in 2026. Whether you are an entrepreneurial founder planning a pilot run or a supply chain executive seeking to consolidate 3PL and FBA fulfillment at Matsun Nutrition, this blueprint provides the strategies required to launch, scale, and legally protect your supplement brand.
Executive Takeaways
- The “Turnkey” IP Trap: Relying on a manufacturer’s in-house R&D without an Intellectual Property Assignment Agreement can result in the factory legally owning your final formulation.
- The Financial Reality of 2,500 MOQs: While industry standards demand 5,000 to 10,000 units, securing a 2,500-unit pilot run limits startup capital risk. Scaling to 10,000 units typically reduces per-unit costs by over 60%.
- Surviving Amazon’s TIC Mandate: To avoid listing suspensions, brands must partner with U.S.-based, cGMP-certified facilities capable of producing ISO 17025-accredited Certificates of Analysis (COAs) that screen for heavy metals and verify label claims.
- The Pharmacokinetics of Liquids: Because liquid formulations bypass the mechanical disintegration and dissolution phases required by traditional solid pills, their active ingredients are immediately available for physiological absorption upon ingestion [2].
- The Uninsured Offshore Liability: Overseas manufacturers’ product liability insurance almost universally excludes the United States, leaving the domestic brand owner solely liable for product recalls or lawsuits [3].
Disclaimer: The regulatory and financial information provided is for educational purposes. Always consult legal counsel for IP agreements and verify current Amazon TIC policies, as they are subject to change.
The State of the Supplement Industry: Growth and Trajectory
The dietary supplement industry is accelerating as aging populations seek well-being and younger demographics prioritize proactive healthcare. To capture this surging demand, agile brands can manage operational costs more effectively by outsourcing production rather than building their own plants. Contract manufacturing allows brands to focus on core strengths while ensuring products meet high-quality standards by leveraging the specialized expertise and advanced equipment of established nutraceutical contract manufacturing organizations (CMOs).
Why the $182 Billion Market Demands Specialized Manufacturing
The global nutraceutical contract manufacturing services market was valued at $182.47 billion in 2024 and is projected to reach $386.6 billion by 2033, representing a robust 8.7% CAGR [1]. The dietary supplement sub-segment shows even more aggressive momentum, expected to grow at a 12.6% CAGR [4].
Building an FDA-compliant facility requires millions in capital and years of meticulous operational setup to adhere strictly to 21 CFR Part 111 guidelines [5]. Leveraging agile contract manufacturing is an essential strategy for both entrepreneurial founders and established supply chain executives to enter and scale quickly.
By offloading facility audits, raw material quarantine, and 3PL fulfillment logistics, brand owners can redirect capital toward customer acquisition while ensuring strict adherence to DSHEA regulations.
The Radical Shift Toward Personalized Nutrition and Clean Labels
The era of generic multivitamins is ending. The personalized nutrition market will reach $37.3 billion by 2032, driven by advanced health tracking, DNA testing, biometric wearables, and consumer demand for targeted wellness results [6]. Modern consumers expect transparency and sophisticated products that avoid artificial excipients or synthetic binders.
To capitalize on this shift, brands must rely on their contract manufacturers’ advanced R&D. An elite manufacturing partner will:
- Engineer clean-label formulations using high-value functional ingredients (like CoQ10 and L-theanine) and natural sweeteners (like stevia).
- Ensure shelf stability for complex biochemical suspensions.
- Verify structure/function claims to ensure active ingredients align with consumer expectations and federal law.
Decoding the Contract Manufacturing Operational Lifecycle
The nutraceutical manufacturing process is incredibly complex. To successfully launch a new product for the market, the standard operational lifecycle includes:
- Ingredient sourcing and raw material quarantine.
- Custom formulations and R&D.
- Pilot batch testing and stability checks.
- Production, final packaging, labeling, and order fulfillment.
Because these steps are so rigorous, choosing the right nutraceutical contract manufacturer is crucial. Whether you are launching rapidly with private label stock formulas or developing complex custom products, your manufacturing partner ultimately dictates your regulatory compliance, speed to market, and overall product excellence.

The Private Label Quality Trap: Cheap Offshore vs. Premium Domestic
Choosing the ‘easy route’ of white-labeling existing, mass-produced formulas from overseas facilities often means selling an inferior, highly commoditized product. The race for the lowest offshore price results in the use of the cheapest ingredients, often loaded with unnecessary synthetic excipients and cheap fillers.
This strategy damages brand reputation and generates negative consumer reviews immediately. Premium domestic private labeling solves this “commodity trap.” Utilizing a U.S.-based manufacturer provides access to proven, high-quality stock formulas produced under strict cGMP compliance.
This lets founders bypass lengthy R&D and achieve rapid 1-to-2-day turnarounds, safely bringing a product to market that legally supports its structure/function claims without sacrificing ingredient integrity.
The Mechanics of Custom R&D, Botanical Sourcing, and Pilot Testing
Custom formulations in nutraceuticals allow brands to target specific health benefits. Rather than simply mixing raw materials, expert manufacturers assist in developing supplement formulas through advanced bench work—perfecting ingredient ratios, masking bitter botanicals, and keeping complex active ingredients (like CoQ10) stable in suspension to maximize bioavailability.
The R&D lifecycle begins with a bench-top prototype to dial in taste and texture. The formula then undergoes rigorous pilot and stability testing to ensure the active compounds won’t degrade once filled into final packaging, such as custom glass or PET bottles.
Elite manufacturers ensure strict ingredient traceability and use comprehensive third-party lab testing to prevent botanical adulteration. Before the product leaves the facility, it is secured with tamper-evident seals and branded shrink sleeves, ensuring the physical product is prepped to survive retail and complex e-commerce supply chains.
The "Turnkey" IP Trap: Guarding Your Most Valuable Asset
At Matsun Nutrition, we’ve seen too many brilliant founders lose control of their brands because a manufacturer buried an IP retention clause in the fine print. That is why our onboarding process includes an explicit Intellectual Property Assignment Agreement before a single pilot batch is mixed. If you bring us a base concept and our R&D team stabilizes the viscosity or perfects the flavoring system, 100% of that finalized commercial formula belongs to you. We are here to scale your business as your manufacturing partner, not to hold your most valuable asset hostage.
A major, often overlooked vulnerability for an emerging supplement brand is the loss of its proprietary intellectual property (IP). Many founders mistakenly believe they legally own the final formulation because they conceptualized the product, provided the initial ingredients, or paid for manufacturing. In reality, the legal aspect of contract manufacturing is more complex. If a facility’s in-house chemists modify or stabilize a recipe without an explicit, pre-existing IP assignment agreement, the factory can legally claim ownership of that revised formula, essentially holding the brand’s most valuable asset hostage.
Why Your Manufacturer Might Legally Own Your Formula (And How to Prevent It)
The trap springs during R&D. A brand provides a base formula, but for the product to remain shelf-stable or palatable, the manufacturer’s chemists must make necessary adjustments, such as:
- Swapping out specific excipients
- Optimizing the suspension of active ingredients
- Refining the flavor profile
Legally, if the manufacturer creates this final, commercial-ready iteration, they claim authorship and ownership of the intellectual property [7]. This traps brands.
If the brand eventually outgrows the manufacturer and attempts to transition production to a larger facility to streamline FBA shipping or 3PL logistics, they suddenly discover they are legally barred from taking their own formula with them. The original manufacturer can either refuse to release the exact formulation sheet or demand an exorbitant, five-figure buyout fee to release the rights.
⚠️ Contractual IP Protection: Why You Need Explicit Assignment Before Pilot Testing A standard NDA isn't enough; it protects confidentiality, not ownership. To prevent disputes, brands must execute a comprehensive Intellectual Property Assignment Agreement before the first bench-top prototype is mixed. This legal document must explicitly state that the brand retains 100% ownership of the final commercial formula, including all modifications, improvements, or stability adjustments made by the manufacturer's staff during the pilot phase. Ethical partners, like Matsun Nutrition, proactively clarify formula ownership by integrating strict IP assignment into their standard contracts to protect clients' proprietary blends and ensure full operational control.
The Financial Reality of Low MOQ Manufacturing
For emerging supplement brands, traditional Minimum Order Quantities (MOQs) of 5,000–10,000 units per SKU tie up vital capital. Nutraceutical contract manufacturing reduces financial risks by offering strategic low MOQs, empowering you to test the market and validate formulations without crippling upfront costs. However, founders must understand how specific delivery formats dictate minimum order requirements and where scaling production improves unit economics.
The "Low MOQ" Illusion: Why Format Dictates Volume (Stick Packs vs. Liquid Bottles)
Your chosen delivery format dictates the required production volume. Complex custom formats like powdered stick packs frequently demand massive minimums (e.g., 100,000+ units) due to extensive machine changeover times and large raw material contracts. By contrast, liquid formulations are far more accessible. Specialized manufacturers have engineered their liquid-filling lines to accommodate smaller, agile batches using standard PET bottles and glass droppers, ensuring fast time-to-market without crippling upfront costs.
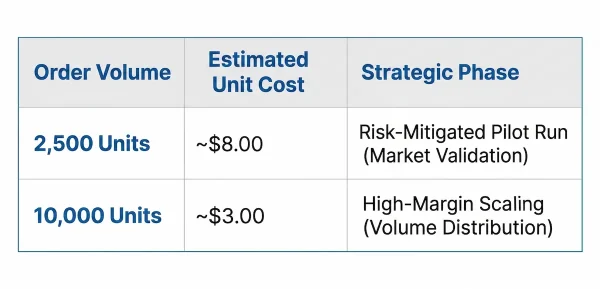
Why is 2,500 Units the Sweet Spot for Low MOQ?
While industry standards typically range from 5,000 to 10,000 units, securing a 2,500-unit pilot run significantly lowers the initial capital risk for emerging brands.
This strategic volume allows founders to test real-world market demand, validate packaging durability during FBA transit, and gather crucial early consumer reviews without incurring excessive inventory costs or expiring inventory. It acts as the ultimate market-validation tool before committing to enterprise-level scale.
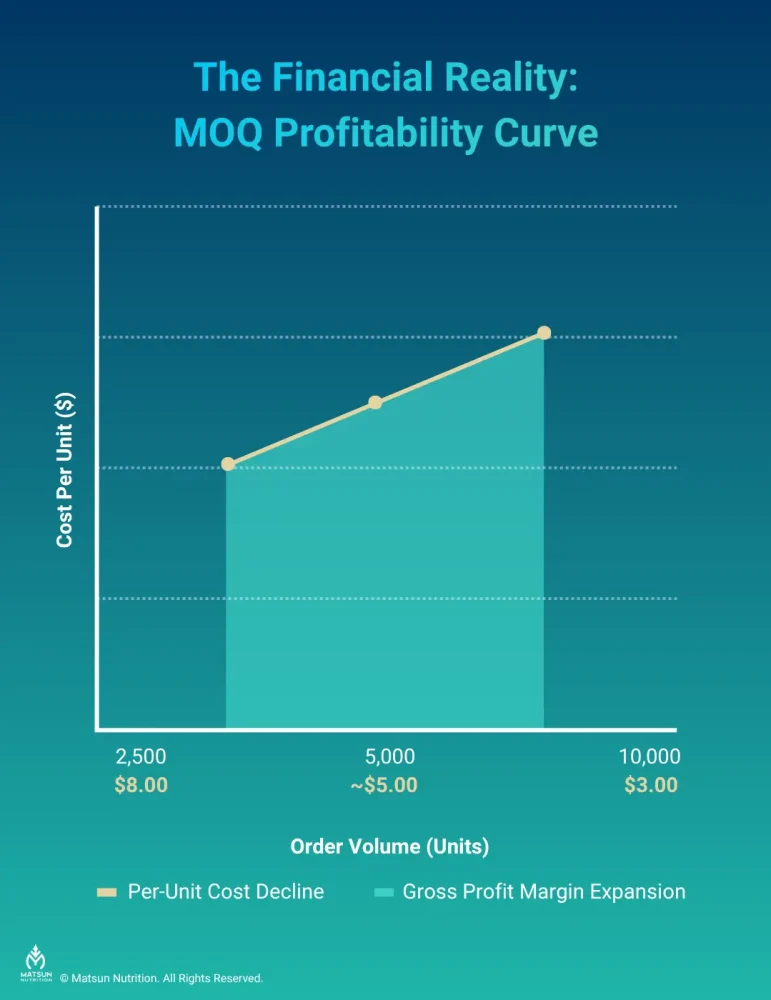
Unit Economics: The Mathematical Tipping Points of Profitability
To visualize the financial impact of scaling production, consider this interactive model of unit costs versus order volume for liquid nutraceuticals. Observe how the unit cost drops between a standard low MOQ and moderate volume, highlighting the critical tipping points. Move the slider or enter a target MOQ above to see the estimated unit cost and required capital investment.
While low MOQs entail a higher initial per-unit cost, unit costs plummet at scale [8]:
Brands should use low MOQs for initial market validation, then use manufacturer scalability to expand gross profit as sales accelerate.
Navigating the Regulatory Minefield: FDA, cGMP, and Legal Liability
The dietary supplement industry faces increasing regulatory oversight, making strict compliance with GMP and FDA standards vital for long-term success. Navigating 21 CFR Part 111 guidelines and Amazon’s new third-party testing mandates requires maintaining rigorous quality control at every stage. By partnering with a fully vetted, domestic manufacturer, brand owners ensure their products comply with regulations, establishing a primary line of defense against the financial and legal threats of contaminated products, recalls, and product liability lawsuits.
The Amazon TIC "Rejection Loop": How Exact COA Wording Saves Your Listing
Effective April 2024, Amazon radically overhauled its dietary supplement policy, essentially weaponizing compliance against unvetted sellers [9]. The platform now requires all sellers to submit ISO 17025-accredited Certificates of Analysis (COAs) through a closed network of approved third-party Testing, Inspection, and Certification (TIC) organizations (such as NSF or Eurofins).
However, generating a COA is not enough. Brands are frequently caught in the “Amazon TIC Rejection Loop,” a frustrating cycle of listing suspensions—because their documentation is not perfectly synchronized. Amazon’s automated systems will reject the submission if the COA does not exactly match the physical product label or the factory’s GMP certificate regarding:
- Active ingredients
- Total plate counts
- Yeast/mold limits
Working with a sophisticated U.S.-based manufacturer ensures these documents are generated the first time correctly, ensuring uninterrupted FBA (Fulfillment by Amazon) and FBM (Fulfillment by Merchant) sales velocity.
(For official guidance on these updates, refer to Amazon Seller Central’s TIC documentation [10].)
The "Uninsured Offshore" Liability: Why Foreign Insurance Policies Leave U.S. Brands Exposed
In an attempt to maximize early profit margins, emerging brands often look to overseas manufacturers. However, this strategy often overlooks a significant legal vulnerability: the “Uninsured Offshore” trap.
While an overseas factory may claim to hold product liability insurance, those foreign policies almost universally exclude the United States from coverage [11]. Furthermore, U.S. courts have limited jurisdiction over foreign entities. If a batch is contaminated with heavy metals (lead, arsenic, cadmium, mercury) and triggers a lawsuit or an FDA recall, the foreign manufacturer is effectively shielded from litigation.
The domestic brand owner is left entirely exposed, assuming 100% of the financial and legal liability. Utilizing a domestic, USA-based manufacturer ensures enforceable legal protection by transferring the liability burden to a partner with valid U.S.-recognized insurance policies.
Delivery Formats: Why Liquid Supplements Scientifically Thrive
While nutraceutical delivery formats include powders, capsules, tablets, and gummies, consumer demand shifts aggressively toward liquids due to superior bioavailability. Bypassing the stomach’s mechanical breakdown, liquid extracts avoid digestive degradation, offering immediate absorption and profound clinical advantages for demographics suffering from dysphagia, while also providing an ideal delivery mechanism for the booming pet health sector.
Why Are Liquid Supplements More Absorbable Than Solid Pills?
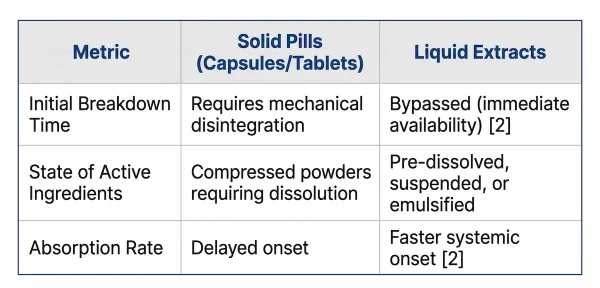
Liquid supplements are significantly more absorbable than solid pills because they completely bypass the stomach’s harsh, time-consuming mechanical breakdown process.
When a consumer ingests a solid pill, the body must expend energy on “digestive dissociation.” Exposing sensitive ingredients to harsh gastric acids severely degrades active nutrients before they can enter the bloodstream. Liquid extracts, however, hold active ingredients in a stable suspension, allowing them to permeate mucous membranes directly and rapidly.
Bioavailability Comparison: Solid vs. Liquid Delivery
Rapid absorption is vital for complex, synergistic stacks like D3 + K2, and for nutrient-dense botanical blends with bladderwrack and burdock root. In liquid form, these compounds are delivered to the bloodstream intact and at peak potency, free from synthetic excipients used to bind solid tablets.
The "Crushed Pill" Dilemma: Solving the Dysphagia Market Gap in Aging Populations
The shift toward liquid formats is not just an optimization; it’s a critical medical necessity for a large segment of the population. Dysphagia, or difficulty swallowing, affects approximately 22% of adults over 50 [12], making traditional pill consumption a major daily hurdle.
In care settings and at home, consumers frequently crush solid supplements to mix into food or water. However, crushing solid medications dangerously alters absorption characteristics and pharmacokinetics, destroying intended time-release profiles and degrading active ingredients before absorption. Liquid supplements—often naturally sweetened with stevia or fruit extracts—provide a vital, safer alternative, ensuring accurate dosing for older demographics.
The Crossover Arbitrage: The Growth of Human-Grade Pet Health Tinctures
The “pet humanization” trend—expected to drive a $4.65 billion pet supplement market by 2033 [13]—offers massive “crossover arbitrage” for agile brands. Since administering solid pills to animals is difficult, brands can use existing human-grade liquid manufacturing infrastructure to produce clean-label, high-efficacy pet health tinctures. By formulating highly palatable options—such as hip-and-joint formulas featuring suspended glucosamine or skin-and-coat drops rich in omega fatty acids—brands can instantly double their addressable product lines with virtually zero R&D friction.
The 7 Non-Negotiable Questions You Must Ask Your Manufacturer
Selecting a contract manufacturer is a critical strategic decision for supplement brands, impacting product quality, legal compliance, and long-term success. Brand owners must ask detailed questions about certifications, testing capabilities, intellectual property rights, and communication to ensure a reliable partnership.

-
What independent, third-party certifications do you actively hold?
Do not accept a simple "FDA registered" claim. Demand proof of active cGMP certification from objective, recognized authorities (NSF International, NPA, or USP).
-
Will you explicitly assign 100% intellectual property (IP) ownership of the final formula to my brand?
Before R&D begins, ensure a written agreement prevents the manufacturer from claiming ownership of any formula modifications, flavor profiles, or stability adjustments they make.
-
What is your true Minimum Order Quantity (MOQ), and what is the exact pricing tier as we scale?
Understand the financial difference between a 2,500-unit pilot run used to mitigate risk versus the profitable unit economics of a 10,000+ unit mass-market order.
-
Do you conduct in-house identity testing (like HPTLC) on all incoming raw botanicals?
Ask how early the testing process starts to ensure raw materials meet purity specifications and are free from adulterants before blending begins.
-
Will you provide the ISO 17025-accredited Certificates of Analysis (COAs) required for Amazon TIC verification?
If selling on Amazon, your manufacturer must supply accurate COAs that seamlessly pass through Amazon's third-party Testing, Inspection, and Certification (TIC) portals.
-
Do you carry product liability insurance that is fully enforceable within the United States?
If vetting an overseas supplier, foreign insurance policies almost universally exclude the U.S., leaving the domestic brand owner solely liable for damages or recalls.
-
What structured processes do you have for daily communication and supply chain transparency?
Lack of communication is a massive pain point; ensure the manufacturer has protocols to immediately alert you to raw material shortages or production delays to avoid out-of-stock scenarios.
Ready to Scale Your Supplement Brand with Confidence?
Navigating the complexities of FDA regulations, Amazon TIC requirements, and custom liquid formulations shouldn’t be a roadblock to your brand’s growth. At Matsun Nutrition, we combine over 30 years of manufacturing excellence with startup-friendly, low MOQs to help you scale profitably and safely.
Whether you need a risk-mitigated 2,500-unit pilot run or are ready for high-margin, enterprise-level volume, our FDA-registered, cGMP-certified U.S. facility is equipped to provide the full-service support you need to protect your intellectual property and dominate the market.
Frequently Asked Questions About Nutraceutical Contract Manufacturing
-
What does "low MOQ" mean in supplement manufacturing?
Minimum Order Quantity (MOQ) dictates the smallest acceptable production run; while the industry standard requires 5,000 to 10,000 units, startup-friendly manufacturers offer low MOQs around 2,500 bottles to help emerging brands test the market and minimize capital risk.
-
Who owns the intellectual property for custom supplement formulas?
Unless you secure an explicit written agreement beforehand, a contract manufacturer can legally claim intellectual property ownership over your custom formula if their chemists modify or stabilize your proprietary blends.
-
How long does the supplement manufacturing process take?
Standard repeat orders typically take 8 to 12 weeks, while a completely new custom formulation requires 12 to 20 weeks to accommodate rigorous research and development (R&D) and stability testing.
-
Are USA-based supplement manufacturers more compliant than overseas facilities?
Yes, domestic manufacturing strictly adheres to FDA regulations and domestic inspections, whereas foreign manufacturers often carry product liability insurance that explicitly excludes U.S. brands from protection during a lawsuit or product recall.
-
What are Amazon's new TIC testing requirements for dietary supplements?
Amazon now mandates that all Certificates of Analysis (COAs) be verified by an approved third-party Testing, Inspection, and Certification (TIC) organization to confirm that active ingredients match label claims and to screen for heavy metals.
-
Are liquid multivitamins more effective than traditional pills?
Liquid extracts bypass the stomach's mechanical breakdown process (disintegration), meaning the active nutrients are pre-dissolved and immediately available for physiological absorption. In pharmacokinetics, this generally results in a higher initial absorption rate and a faster onset of action compared to traditional capsules or tablets [2].
References
- Straits Research. (2024). Nutraceutical Contract Manufacturing Services Market Size, Demand, Growth Analysis by 2033. Straits Research Industry Reports. https://straitsresearch.com/report/nutraceutical-contract-manufacturing-services-market
- Mansoor, A., & Tollamadugu, V. S. (2023). Drug Absorption. StatPearls Publishing. National Center for Biotechnology Information (NCBI) Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK557405/
- The Coyle Group. (2023). Product Liability Insurance for Importers: What You Need to Know. Commercial Insurance Risk Management. https://thecoylegroup.com/product-liability-insurance-for-importers-what-you-need-to-know/
- Grand View Research. (2024). Dietary Supplements Contract Manufacturing Market Report, 2030. Grand View Research Industry Analysis. https://www.grandviewresearch.com/industry-analysis/dietary-supplement-contract-manufacturing-market-report
- U.S. Food and Drug Administration (FDA). (Current). 21 CFR Part 111 – Current Good Manufacturing Practice in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements. Electronic Code of Federal Regulations (eCFR). https://www.ecfr.gov/current/title-21/chapter-I/subchapter-B/part-111
- Valuates Reports. (2022). Personalized Nutrition Market To Reach USD 37.3 Billion By 2030. PR Newswire. https://www.prnewswire.com/news-releases/personalized-nutrition-market-to-reach-usd-37-3-billion-by-2030–growing-at-a-cagr-of-11-48–valuates-reports-301697883.html
- EPGD Business Law. (2024). Your Product Formula: Who Really Owns It? EPGD Corporate & Business Law. https://www.epgdlaw.com/who-really-owns-your-product-formula/
- Greenlaw, S. A., & Shapiro, D. (2022). The Structure of Costs in the Long Run. In Principles of Microeconomics 3e. OpenStax, Rice University. https://openstax.org/books/principles-economics-3e/pages/7-3-costs-in-the-short-run
- NSF International. (2024). Amazon’s New Dietary Supplements Policy: Enhancing Safety and Compliance. National Sanitation Foundation Knowledge Library. https://www.nsf.org/knowledge-library/amazon-new-dietary-supplements-policy-enhancing-safety-compliance
- Amazon Seller Central. (2024). Dietary Supplements: Our Policy for Dietary Supplements (Testing, Inspection, and Certification). Amazon.com, Inc. https://sellercentral.amazon.com/gp/help/external/55N3JF2WQS7RVNE
- Alston & Bird LLP. (2019). Foreign Manufacturers vs. US Distributors: Contractual Indemnity and Insurance Coverage Governing Product Liability. Alston & Bird Legal Insights. https://www.alston.com/en/insights/publications/2019/09/chinese-manufacturers-vs-us-distributors
- Aslam, M., & Vaezi, M. F. (2013). Dysphagia in the Elderly. Gastroenterology & Hepatology, 9(12), 784–795. National Institutes of Health (NIH). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3999993/
- Grand View Research. (2024). Pet Supplements Market Size, Share & Trends Analysis Report, 2033. Grand View Research Database.https://www.grandviewresearch.com/industry-analysis/pet-supplements-market




